Description
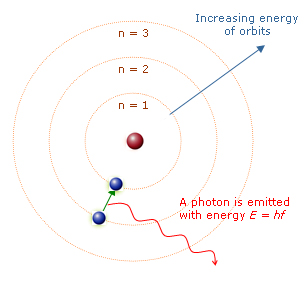
The Bohr atomic model, proposed by Danish physicist Niels Bohr in 1913, is a planetary model where negatively charged electrons orbit a small, positively charged nucleus in fixed, quantized circular paths called orbits or shells. Key principles include quantized electron energy levels, stable non-radiating orbits, and photon emission/absorption during orbital jumps. It specifically explains the hydrogen emission spectrum. While superseded by quantum mechanical models, the Bohr diagram remains one of the most recognizable and widely used scientific visualizations — a powerful example of how a simplified visual model can communicate complex physics. Bohr received the Nobel Prize in Physics in 1922 for this work.